09
2026
-
04
Dalian Institute of Chemical Physics Unveils a New Mechanism for the Photooxidation of Myrcene Leading to Secondary Organic Aerosol Formation Using the Dalian Coherent Light Source
Author:
Recently, a research team led by Researcher Jiang Ling and Associate Researcher Li Gang from the Cluster Spectroscopy and Dynamics Group (Group 2506) of the State Key Laboratory of Chemical Reaction Dynamics at the Dalian Institute of Chemical Physics has employed an independently developed aerosol mass spectrometry method based on the Dalian Coherent Light Source to investigate the photooxidation of β-linalool. Their study has elucidated the mechanistic role of nitrogen oxides (NOx) in this process and their synergistic effects with sulfur dioxide (SO2), which is expected to provide critical scientific evidence for understanding the formation mechanisms of atmospheric pollution and for informing pollution-control policy-making.
Atmospheric particulate pollutants pose serious risks to human health and the ecological environment. According to the report “Air China 2025: Best Practices in China’s Air Pollution Prevention and Control,” although China’s annual average PM2.5 concentration has now met the national Class II air quality standard (PM2.5 ≤ 35 μg/m³), it remains significantly higher than the World Health Organization’s guideline value (PM2.5 ≤ 5 μg/m³). The transformation of gaseous precursors into aerosols is a critical step in the formation of atmospheric particulate pollutants; therefore, precise measurement of aerosol chemical composition and elucidation of their nucleation mechanisms are of great scientific importance for understanding the formation processes of these pollutants and for developing effective prevention and control strategies. As key targets in atmospheric pollution control, NOx (NOx = NO₂ + NO) and SO₂ urgently require clarification of their mechanisms of action in the formation of secondary organic aerosols from volatile organic compounds.
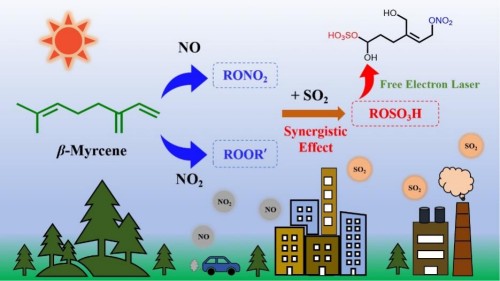
In this study, the teams led by Jiang Ling and Li Gang utilized an aerosol mass spectrometry experimental station based on the Dalian Coherent Light Source to investigate the photooxidation of β-linalool—a typical linear monoterpenoid volatile organic compound—under conditions of NOx and SO2. The results demonstrate that NO2 promotes particle formation and growth by supplying ample oxidants (O3), thereby favoring the production of highly oxidized products; in contrast, NO more readily participates in oxidation reactions, influencing the oxidation pathways of peroxy radicals and enhancing the formation of highly volatile products while simultaneously suppressing the formation of particulate-phase products. Upon the introduction of SO2 into the system, the yields of secondary organic aerosols under both NO2 and NO conditions increased markedly, indicating a synergistic enhancement effect of SO2 and NOx on the photooxidation of β-linalool. Leveraging the extreme ultraviolet single-photon soft-ionization capabilities of the Dalian Coherent Light Source, the team identified a series of novel compounds—including organic peroxides, organic nitrates, and organic sulfates—and, combined with quantum-chemical theoretical calculations, elucidated their formation mechanisms: under NO2 conditions, cross-reactions give rise to highly oxidized products; under NO conditions, peroxy radicals combine with NO to form organic nitrates; and SO2, whether in the presence of NO2 or NO, can induce the formation of new organic sulfate species. These findings thus reveal, at the molecular level, the mechanistic basis for the synergistic interaction between SO2 and NOx. This research not only provides critical experimental and theoretical foundations for improving the predictive accuracy of atmospheric models but also holds promise as a scientific basis for developing regional strategies to control complex pollution.
The relevant research findings, titled “Distinct Roles of NO2 versus NO and Synergisms with SO2 in Secondary Organic Aerosol Formation from β-Myrcene Photooxidation,” were recently published in Environmental Science & Technology. The first author of this work is Zhao Ya, a doctoral student jointly trained by Group 2506 of our institute and the School of Chemistry at Dalian University of Technology. This research was supported by the National Natural Science Foundation of China, the National Natural Science Foundation’s “Frontier Research on Dynamic Chemistry” Science Center project, the Ministry of Science and Technology’s Major Project on Scientific and Technological Innovation 2030, and the Chinese Academy of Sciences’ Category B Pioneer Initiative on “Precision Detection of Chemical Reaction Transition States Based on Extreme Ultraviolet Light Sources,” among other funding programs.
Source: Dalian Institute of Chemical Physics
Previous
Previous

